_ANTIBODY
_Antibodies are immunoglobulins that react specifically with the antigen that stimulated their production. Antibody
is produced by B cell and is present on its membrane. Antibody is
produced and secreted by plasma cells in humoral immune response due to activation of B cell by antigen. Antibody has
the ability to combine specifically with the
antigen that stimulated its production.
All antibodies are immunoglobulins but all immunoglobulins are not antibodies. About 20% of plasma proteins
are immunoglobulins. Antiserum is serum containing antibody.
Structure
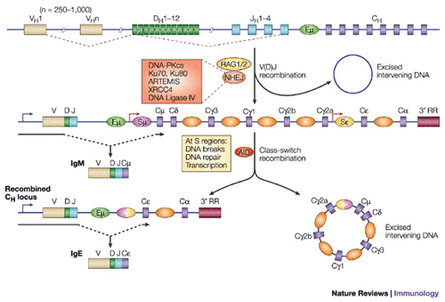
Immunoglobulins are glycoproteins made up of heavy (H) and light (L) polypeptide chains. The terms 'heavy' and 'light' refer to the molecular weight: heavy chains have a molecular weight of about 50,000 and light chains have a molecular weight of about 25,000. All antibody or immunoglobulin molecules consist of one or more basic monomer molecule of four polypeptide chains. A basic monomer molecule (Fig. 63.4) is Y shaped and always consists of two identical heavy (H) polypeptide chains and two identical light (L) polypeptide chains held together by disulphide bonds. Molecular formula of a mon&ner is H2L2. Both H and L chains are subdivided into a variable (V) region at the amino terminal end, and the rest into constant (C) regions.
H chain consists of one variable NO and 3 or 4 constant (CH) regions. L chain consists of one variable NO and one constant ( CL) region. Within variable regions of both light and heavy chains there are three hypervariable regions which constitute the antigen binding site, or complementarily-determining region (CDR). Only 5 to 10 aminoacids in each hypervariable region constitute the antigen-binding site. Antigen binding is noncovalent, involving vander Waals, electrostatic and other weak forces as well as hydrogen and hydrophobic bonds.
Due to antigenic differences in constant regions there are five types of heavy chains designated as y (gamma), p (mu), a (alpha), E; (epsilon) and 6 (delta) producing five classes of immunoglobulins termed as IgG, IgM, IgA, IgE and IgD respectively.
If one monomeric immunoglobulin molecule is treated with the enzyme papain, peptide bonds in the hinge region are broken into two identical Fab (Fragment antigen binding) fragments which carry the antigen binding sites, and one Fc: fragment (Fragment crystallizable) which is involved in placental transfer, complement fixation, attachment of various cells and other biological activities.
Antibody is most effective against extracellular antigens.
Structure
Immunoglobulins are glycoproteins made up of heavy (H) and light (L) polypeptide chains. The terms 'heavy' and 'light' refer to the molecular weight: heavy chains have a molecular weight of about 50,000 and light chains have a molecular weight of about 25,000. All antibody or immunoglobulin molecules consist of one or more basic monomer molecule of four polypeptide chains. A basic monomer molecule (Fig. 63.4) is Y shaped and always consists of two identical heavy (H) polypeptide chains and two identical light (L) polypeptide chains held together by disulphide bonds. Molecular formula of a mon&ner is H2L2. Both H and L chains are subdivided into a variable (V) region at the amino terminal end, and the rest into constant (C) regions.
H chain consists of one variable NO and 3 or 4 constant (CH) regions. L chain consists of one variable NO and one constant ( CL) region. Within variable regions of both light and heavy chains there are three hypervariable regions which constitute the antigen binding site, or complementarily-determining region (CDR). Only 5 to 10 aminoacids in each hypervariable region constitute the antigen-binding site. Antigen binding is noncovalent, involving vander Waals, electrostatic and other weak forces as well as hydrogen and hydrophobic bonds.
Due to antigenic differences in constant regions there are five types of heavy chains designated as y (gamma), p (mu), a (alpha), E; (epsilon) and 6 (delta) producing five classes of immunoglobulins termed as IgG, IgM, IgA, IgE and IgD respectively.
If one monomeric immunoglobulin molecule is treated with the enzyme papain, peptide bonds in the hinge region are broken into two identical Fab (Fragment antigen binding) fragments which carry the antigen binding sites, and one Fc: fragment (Fragment crystallizable) which is involved in placental transfer, complement fixation, attachment of various cells and other biological activities.
Antibody is most effective against extracellular antigens.
_ Immunoglobulin (19) Classes
Immunoglobulins are of 5 types- IgG, IgM, IgA, IgE and IgD.
IgG
Heavy chain symbol is y. Molecular formula H2L2. There are four subclasses IgG1, IgG2, IgG3 and IgG4. Molecular weight is 150,000. Serum concentration is approximately 1000 mg/dl. IgG is 75 per cent of total serum immunoglobulin.
Functions :
1. IgG is the predominant antibody in secondary humoral immune response.
2. Antibody is most effective against extracellular microorganisms.
3. IgG is the only antibody that cross the placenta as syncitiotroph ob last cells have Fc receptor for IgG.
4. Neutralizes bacterial exotoxins e.g. exotoxins of C. diphtheriae, Clostridium tetani are neutralized by specific IgG antibodies.
5. Neutralizes viruses. Antibodies can bind to viruses and limit the establishment of viral infection i.e. to invade host cells, or can prevent further dissemination in the body.
6. IgG acts as opsonin in phagocytosis.
7. Complement activation by classic pathway by antigen- IgG complex.
IgM
Heavy chain symbol is p. Each free serum IgM molecule is composed of five H2L2 units, i.e. a pentamer and consists of five monomer units, each of which is like one IgG molecule (Fig. 63.4), and one J (Joining) chain. Molecular weight is 900,000. Serum concentration is approximately 120 mg/dl. It is 9% of total serum Ig
Functions :
1. Monomer IgM is present on B cell surface and is the specific receptor for antigen.
2. Antigen-IgM complex activates complement by the classic pathway.
3. It is the main immunoglobulin produced in the primary humoral immune response.
4. It is important in defence against bacteria and viruses.
5. It is important in antigen-antibody reactions (Laboratory methods like agglutination, complement fixation).
Immunoglobulins are of 5 types- IgG, IgM, IgA, IgE and IgD.
IgG
Heavy chain symbol is y. Molecular formula H2L2. There are four subclasses IgG1, IgG2, IgG3 and IgG4. Molecular weight is 150,000. Serum concentration is approximately 1000 mg/dl. IgG is 75 per cent of total serum immunoglobulin.
Functions :
1. IgG is the predominant antibody in secondary humoral immune response.
2. Antibody is most effective against extracellular microorganisms.
3. IgG is the only antibody that cross the placenta as syncitiotroph ob last cells have Fc receptor for IgG.
4. Neutralizes bacterial exotoxins e.g. exotoxins of C. diphtheriae, Clostridium tetani are neutralized by specific IgG antibodies.
5. Neutralizes viruses. Antibodies can bind to viruses and limit the establishment of viral infection i.e. to invade host cells, or can prevent further dissemination in the body.
6. IgG acts as opsonin in phagocytosis.
7. Complement activation by classic pathway by antigen- IgG complex.
IgM
Heavy chain symbol is p. Each free serum IgM molecule is composed of five H2L2 units, i.e. a pentamer and consists of five monomer units, each of which is like one IgG molecule (Fig. 63.4), and one J (Joining) chain. Molecular weight is 900,000. Serum concentration is approximately 120 mg/dl. It is 9% of total serum Ig
Functions :
1. Monomer IgM is present on B cell surface and is the specific receptor for antigen.
2. Antigen-IgM complex activates complement by the classic pathway.
3. It is the main immunoglobulin produced in the primary humoral immune response.
4. It is important in defence against bacteria and viruses.
5. It is important in antigen-antibody reactions (Laboratory methods like agglutination, complement fixation).
IgA
Heavy chain symbol is a. IgA may be :
(1) Serum IgA. Monomer (80%) and dimer with J chain. Molecular weight is 170,000. Serum concentration is approximately 200 mg per dl. It is 15 percent of total serum 1g. There are two subclasses- IgA1 and IgA2.
(2) Secretory IgA. It is a dimer consisting of two monomer units (two 1-121-2 units), one J chain and one secretory component. Secretory component is synthesized by mucosal epithelial cells that makes IgA to cross the mucosa. Molecular weight is 400,000.
Functions :
1. It is the main Ig in secretions of respiratory, intestinal and genital tracts, milk, saliva and tear. It prevents attachment of bacteria and viruses to mucous membrane. Some bacteria (e.g. Neisseria) can destroy IgAl by its protease.
2. Aggregated IgA activates the alternate pathway of complement.
IgE
Heavy chain symbol is E. IgE molecule is a monomer (1-121-2) Molecular weight is 190,000. Serum concentration is about 0.05 mg per dl. It is less than 1 percent of total serum Ig.
Functions :
1. It is protective against helminthic infection.
2. Fc region of IgE binds to the surface of mast cells and basophils in type I immediate hypersensitivity.
IgD
It has no antibody activity. It acts as antigen receptor on surface of many B cells.
_
Heavy chain symbol is a. IgA may be :
(1) Serum IgA. Monomer (80%) and dimer with J chain. Molecular weight is 170,000. Serum concentration is approximately 200 mg per dl. It is 15 percent of total serum 1g. There are two subclasses- IgA1 and IgA2.
(2) Secretory IgA. It is a dimer consisting of two monomer units (two 1-121-2 units), one J chain and one secretory component. Secretory component is synthesized by mucosal epithelial cells that makes IgA to cross the mucosa. Molecular weight is 400,000.
Functions :
1. It is the main Ig in secretions of respiratory, intestinal and genital tracts, milk, saliva and tear. It prevents attachment of bacteria and viruses to mucous membrane. Some bacteria (e.g. Neisseria) can destroy IgAl by its protease.
2. Aggregated IgA activates the alternate pathway of complement.
IgE
Heavy chain symbol is E. IgE molecule is a monomer (1-121-2) Molecular weight is 190,000. Serum concentration is about 0.05 mg per dl. It is less than 1 percent of total serum Ig.
Functions :
1. It is protective against helminthic infection.
2. Fc region of IgE binds to the surface of mast cells and basophils in type I immediate hypersensitivity.
IgD
It has no antibody activity. It acts as antigen receptor on surface of many B cells.
_